Strategic Insights for Industry Leaders
Entering the Pharmaceutical and Healthcare Market in Malaysia
Introduction
Pharmaceutical and healthcare companies exploring Southeast Asia increasingly identify Malaysia as a strategic entry point due to its strong regulatory framework, well-developed healthcare infrastructure, and growing demand for specialised medical solutions. As regional healthcare needs evolve and competition intensifies, Malaysia offers significant opportunities for organisations seeking sustainable market expansion and long-term commercial growth.
However, entering the Malaysian pharmaceutical and healthcare market requires more than recognising its potential. Companies must navigate regulatory approvals, pricing structures, stakeholder expectations, and complex market dynamics while building strong local partnerships and sustainable commercial strategies. For pharmaceutical organisations planning to establish a presence in the Malaysian pharmaceutical sector, a structured and informed market-entry approach is essential to ensure successful and compliant operations.
Why Malaysia is a Strategic Pharmaceutical Market
Malaysia’s pharmaceutical and healthcare sector has experienced steady and consistent growth over the past decade, supported by government initiatives, increasing healthcare expenditure, and rising demand for specialised treatments. The country’s dual healthcare system, consisting of both public and private sectors, provides broad market access while allowing premium and specialised healthcare services to flourish.
It is projected to reach approximately USD 1.7-2.2 billion by 2030, supported by steady growth in healthcare expenditure, rising chronic disease burden, and increasing demand for specialised therapies and healthcare products in Malaysia.
Malaysia’s strong regulatory governance, combined with a growing private healthcare sector and increasing patient awareness, creates an environment where specialised healthcare solutions, including advanced reproductive treatments, medical aesthetic innovations, and medical nutrition support, can be introduced and scaled with the right strategic approach.
Understanding the Local Healthcare Ecosystem
A clear understanding of Malaysia’s healthcare ecosystem is fundamental for successful market entry. The market is shaped by a complex network of stakeholders, including regulators, healthcare professionals, distributors, hospitals, clinics, and private healthcare institutions, all of which influence product adoption and commercial success.

It is also worthwhile to note that Malaysia is recognised as a trusted destination for medical tourism, attracting patients from China, Indonesia, and the Middle East. This reinforces the country’s position as a regional hub for advanced treatments and innovative healthcare solutions, further strengthening demand for high-quality pharmaceutical and specialised medical offerings.
Malaysia’s public healthcare system focuses on accessibility and affordability, while the private sector drives specialised treatments, innovative therapies, and premium healthcare services. Both systems often adopt new technologies and clinical innovations, particularly in areas such as reproductive health, medical aesthetic procedures, and advanced therapeutic solutions. Pharmaceutical companies must therefore develop strategies that address both sectors while aligning with regulatory and pricing expectations.
Successful market entry requires collaboration with established local pharmaceutical distributors, healthcare institutions, and clinical networks to ensure efficient product availability and accessibility. Distribution strategies must go beyond logistics, incorporating market insight, stakeholder alignment, and regulatory compliance to support sustainable penetration and long-term growth.
Healthcare professionals, including physicians, specialists, pharmacists (both independent and retail), and clinical practitioners, play a central role in shaping product adoption and treatment decisions. Building credibility through scientific communication, education, and professional engagement with these healthcare professionals is therefore essential, particularly for innovative therapies where clinical understanding and trust are critical for adoption.
Understanding how these components interact enables pharmaceutical companies in Malaysia to position their healthcare products effectively and align their commercial strategies with real market needs.
Regulatory and Market Entry Considerations
Regulatory compliance is one of the most critical aspects of entering Malaysia’s pharmaceutical and healthcare market. Companies must ensure that all healthcare products in Malaysia meet strict safety, quality, and regulatory standards before commercialisation.

Companies must secure product registration with the National Pharmaceutical Regulatory Agency (NPRA) for pharmaceuticals, while medical devices are registered with the Medical Device Authority (MDA). NPRA is also involved in cases involving medical devices classified as combination products. Understanding the appropriate regulatory pathway early in the process helps prevent delays and ensures smoother market entry.
Organisations must align early with pricing expectations, reimbursement landscape, and tender dynamics across both public and private sectors. A clear understanding of procurement processes and pricing structures is essential to ensure sustainable commercialisation and competitive positioning.
Companies must adhere to local regulations, including pharmacovigilance, labelling standards, and promotional compliance. Maintaining regulatory discipline and operational transparency is essential to ensure long-term credibility and uninterrupted market operations.
A successful entry in the Malaysian market requires pharmaceutical companies to partner with a high-calibre and established local distributor that combines strong marketing capabilities with deep local insight, ensuring not just a successful product launch, but sustained brand growth, resilience against market shifts, and the agility to navigate an increasingly fast-evolving healthcare landscape.
Market Entry Framework for Pharmaceutical Companies Interested in Entering Malaysia
For organisations planning to enter the Malaysian market, a structured framework helps reduce risk and improve execution efficiency. A strategic market entry approach typically involves five key stages.
Companies must first evaluate therapeutic demand, competitive landscape, pricing structures, and healthcare access pathways to determine market viability and long-term potential.
A clear regulatory roadmap ensures that pharmaceutical and healthcare products meet approval requirements and can be introduced efficiently without delays.
Choosing an established local partner is critical to ensure market access, stakeholder engagement, strategic brand positioning, and successful commercialisation of products.
Building relationships with healthcare professionals, clinical institutions, and distribution networks strengthens product credibility and adoption.
Successful market entry requires coordinated marketing, scientific communication, and commercial execution to build long-term market presence.
This structured approach enables pharmaceutical companies in Malaysia and new entrants to establish a strong and sustainable foundation.
Common Market Entry Mistakes in Malaysia
Despite strong market potential, many pharmaceutical and healthcare organisations encounter challenges due to strategic misalignment or insufficient local understanding.
Market access is not driven by a single stakeholder. Success requires navigating regulators, payors, hospital administrators, pharmacists, and prescribers, each with different priorities.
In Malaysia’s dispensing environment, product uptake depends on balancing prescriber intent with pharmacist substitution, availability, and commercial considerations, requiring precise stakeholder and channel alignment.
Different procurement models, pricing expectations, and access pathways between public and private sectors create execution complexity for new entrants.
Evolving regulatory requirements, local compliance standards, and approval timelines can delay entry and impact launch momentum if not proactively managed.
Reliance on local partners, inventory planning, and demand variability can create inefficiencies without strong in-market coordination.
Limited awareness, lack of clinical familiarity, and absence of established networks slow product adoption, especially in competitive or mature therapeutic areas.
Organisations that proactively address these challenges are more likely to achieve sustainable growth and long-term market success in Malaysia.
Strategic Commercialisation Partner vs Traditional Distribution Model
When entering Malaysia, pharmaceutical companies often evaluate different commercialisation approaches. Beyond market access, the choice of a local partner has a direct impact on resource deployment, stakeholder engagement, and long-term value creation.
Traditional distributor models typically operate with broad portfolios, where sales teams manage multiple products across categories, often limiting focus and depth of engagement. In contrast, a strategic commercialisation partner deploys dedicated, focused resources aligned to specific brands and therapeutic areas, enabling more targeted execution, stronger stakeholder relationships, and sustained market development.
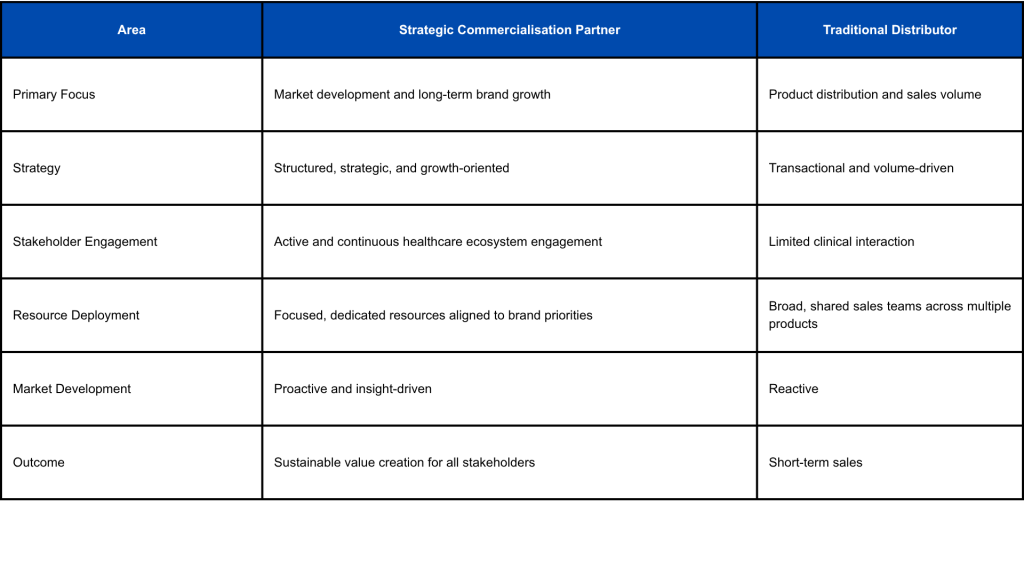
A strategic commercialisation approach supports pharmaceutical companies in building long-term credibility, particularly in specialised healthcare areas where clinical education, patient awareness, innovation, and regulatory compliance play a significant role in market adoption.
Malaysia Market Outlook: Why the Right Partner Defines Success
Malaysia’s pharmaceutical and healthcare sector continues to present strong long-term opportunities, but success increasingly depends on strategic execution, continuous innovation, and strong local partnerships.

Rising chronic disease burden, an ageing population, and increasing health awareness are driving demand for innovative therapies, advanced treatment modalities, and specialised healthcare solutions.
Malaysia serves as a strong foundation for regional growth, allowing companies to test and scale their strategies across Southeast Asia.
Healthcare stakeholders now expect not just product availability, but differentiated value, clinical outcomes, education, and long-term brand trust, particularly for innovative and specialised therapies.
Successful companies go beyond access, actively shaping demand through clinical engagement, patient pathways, innovation adoption, and ecosystem influence rather than relying solely on distribution reach.
With the increasing presence of global and regional players, companies without a focused strategy risk being commoditised, deprioritised, or overshadowed within broad distributor portfolios. Continuous innovation and strategic positioning are therefore essential to remain competitive.
Should Malaysia Be Part of Your Growth Trajectory?
Entering the Malaysian pharmaceutical and healthcare market requires a structured, strategic, and locally informed approach. Regulatory compliance, stakeholder engagement, pricing alignment, and strong commercial execution all play critical roles in determining long-term success.
In an increasingly complex and competitive environment, companies benefit from working with established local partners who understand Malaysia’s healthcare ecosystem, regulatory landscape, and commercial dynamics. A strategic commercialisation partner bridges the gap between market entry and sustainable brand growth, ensuring that pharmaceutical and healthcare products are positioned effectively and supported by strong stakeholder engagement.
As the market continues to evolve, innovation will be a key differentiator. Companies must not only introduce new therapies and solutions, but also work with partners who can support the ethical, compliant, and effective adoption of innovation within the healthcare ecosystem.
With deep local market understanding, strong healthcare ecosystem expertise, and a proven ability to support specialised healthcare segments, including reproductive health, medical aesthetic solutions, and medical nutrition support, organisations such as EP Plus Group are well-positioned to help pharmaceutical and healthcare companies navigate Malaysia’s evolving market landscape and build sustainable long-term success.
To learn more, visit www.epplusgroup.com